Therapeutic Equivalence Explained: Is Your Generic Drug Safe?

You've probably been there: you go to the pharmacy to pick up a prescription, and the pharmacist tells you they've substituted your brand-name medication for a generic version. Most of us just nod and take the cheaper option, but a crucial question often lingers: is this new pill actually doing the exact same thing in my body? This is where therapeutic equivalence is the scientific determination that two drug products with the same active ingredients will produce the same clinical effect and safety profile . It is the invisible safety net that ensures a cost-saving switch doesn't lead to a medical mishap.
The Three Pillars of Drug Sameness
For a drug to be labeled as therapeutically equivalent, it can't just "look" like the original. It has to pass three rigorous scientific tests. First, there is pharmaceutical equivalence. This means the generic must have the exact same active ingredient, the same strength, the same dosage form (like a tablet or capsule), and the same way of entering the body. While the filler ingredients, color, or shape might change, the "engine" of the drug must be identical.
Second is bioequivalence. This is where the FDA gets technical. They don't just check if the drug is there; they check how it behaves. Bioequivalence proves that the rate and extent of absorption of the generic drug don't differ significantly from the brand name. Specifically, the FDA requires that the drug's concentration in the blood stays within a tight window-typically 80% to 125% of the brand-name drug's performance. If a drug absorbs too fast or too slow, it could be ineffective or even toxic.
Finally, these two factors must lead to an identical clinical effect. If the drug is pharmaceutically identical and bioequivalent, it is expected to have the same safety profile and therapeutic outcome when administered to patients under the conditions specified in the labeling.
Decoding the Orange Book
How do doctors and pharmacists actually verify this? They use a massive database called the Orange Book, managed by the FDA's Center for Drug Evaluation and Research. This isn't just a list; it's a grading system. If you see a two-letter code like "AB" next to a drug, it's a green light. An "A" rating means the FDA failed to find any evidence of therapeutic inequivalence, and "B" refers to specific product details. In short, an "AB" rating means the generic is a safe, direct substitute.
| Criteria | Pharmaceutical Equivalence | Therapeutic Equivalence |
|---|---|---|
| Active Ingredient | Identical | Identical |
| Dosage Form | Identical | Identical |
| Absorption Rate | Not necessarily tested | Must be Bioequivalent (80-125% range) |
| Clinical Outcome | Not guaranteed | Expected to be identical |
When "Close Enough" Isn't Enough: Narrow Therapeutic Index Drugs
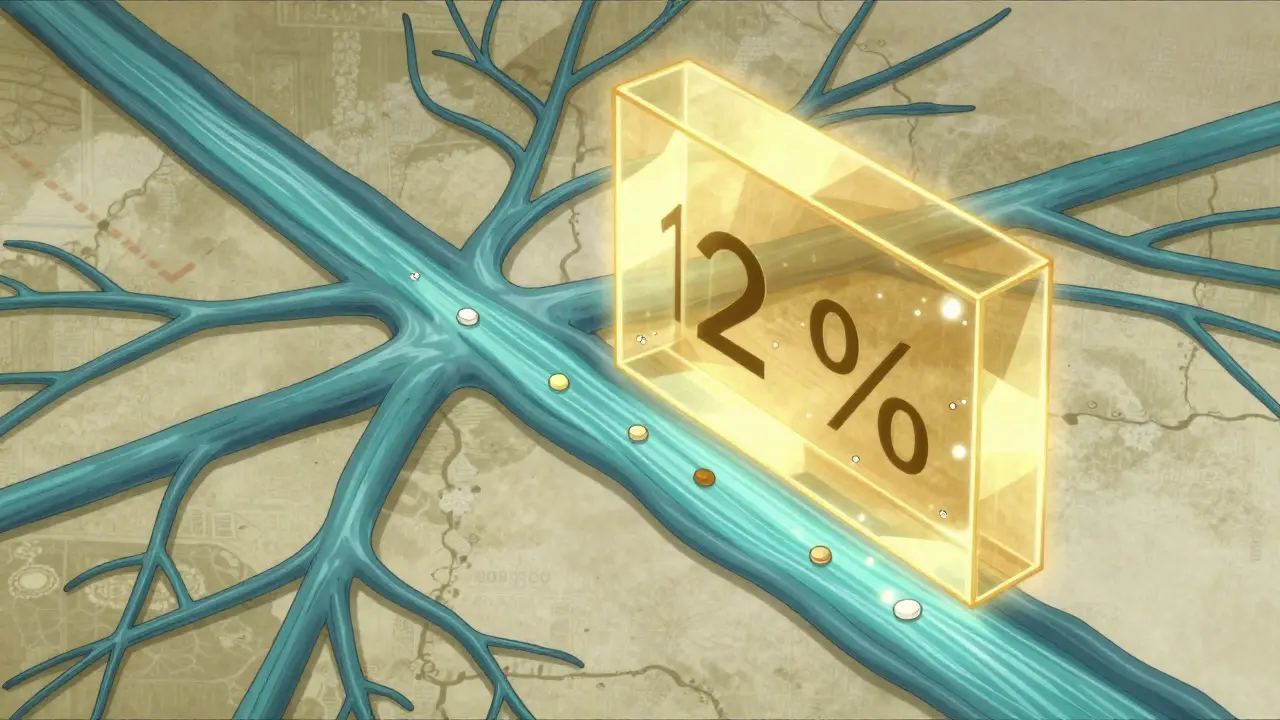
For most medications, a small variation in absorption doesn't matter. But for some, it's a huge deal. These are called narrow therapeutic index (NTI) drugs. Think of drugs like warfarin (a blood thinner) or levothyroxine (for thyroid health). With NTI drugs, the gap between a dose that works and a dose that is toxic is tiny. Even a 10% difference in how a drug is absorbed could lead to a patient bleeding too much or having an underactive thyroid.
Because of this, the FDA applies much stricter rules to NTI drugs. Instead of the standard 80-125% bioequivalence window, they often require a much tighter 90-110% confidence interval. This ensures that the generic is almost a mirror image of the brand name, removing the risk of clinical instability for sensitive patients.
Generic Substitution vs. Therapeutic Interchange
There is a common mistake people make: confusing generic substitution with therapeutic interchange. They sound similar, but one is significantly riskier than the other. Generic substitution happens when a pharmacist replaces a brand-name drug with a therapeutically equivalent generic (the "AB" rated ones). This is generally very safe and, according to some data, has helped healthcare systems save trillions of dollars without compromising care.
Therapeutic interchange, however, is different. This is when a doctor or pharmacist switches a patient to a different drug within the same class. For example, switching one cholesterol medication for another similar one that isn't the same molecule. While this is sometimes necessary, it carries more risk. In fact, a 2019 study in JAMA Internal Medicine found that adverse event reporting was 32% higher with therapeutic interchange compared to simple generic substitution. Stick to the therapeutically equivalent options whenever possible.

The Challenge of Complex Generics
Not every drug is a simple pill. Inhalers for asthma or topical steroid creams are far more complex to replicate. The FDA acknowledges that traditional bioequivalence tests-which mostly look at blood levels-don't always tell the whole story for these products. If a cream doesn't penetrate the skin correctly, it doesn't matter if the active ingredient is the same; it won't work.
To fix this, the FDA is currently updating its guidance for "complex generics." They are moving toward more sophisticated testing and even exploring artificial intelligence to predict how different formulations might affect a patient's outcome. They've partnered with MIT to build machine learning models that can flag potential issues in a drug's formulation before it even hits the market.
What This Means for Your Next Pharmacy Visit
If you're feeling anxious about a switch, remember that the system is designed to protect you. Most patients-around 87% according to UnitedHealthcare data-report identical effects when switching to an Orange Book-listed generic. However, if you are on an NTI medication or a complex inhaler, it is always worth asking your pharmacist: "Is this an AB-rated therapeutic equivalent?"
Knowing the difference allows you to enjoy the cost benefits of generics while keeping your safety as the top priority. If you have a history of reacting poorly to different fillers (inactive ingredients), tell your provider. While the active drug is the same, the dyes or binders can occasionally cause issues for hypersensitive individuals.
What is the difference between a generic and a therapeutically equivalent drug?
A generic drug is any medication created to be similar to a brand-name drug. A therapeutically equivalent drug is a specific type of generic that has been scientifically proven to have the same active ingredient, the same dosage, and the same absorption rate (bioequivalence), ensuring it produces the same clinical result as the brand name.
Can I trust a generic drug as much as a brand-name drug?
Yes, provided it is listed as therapeutically equivalent (such as an "AB" rating in the FDA Orange Book). These drugs must meet strict standards for purity, potency, and performance, meaning they are expected to work exactly like the original.
Why do some people feel differently after switching to a generic?
While the active ingredient is the same, generic drugs use different inactive ingredients (fillers, binders, and dyes). Some patients may have sensitivities to these specific additives, or they may experience a "nocebo" effect, where the psychological expectation that a generic is inferior leads to perceived side effects.
What are Narrow Therapeutic Index (NTI) drugs?
NTI drugs are medications where a very small change in the blood concentration can lead to a significant change in the drug's effect, potentially leading to toxicity or treatment failure. Examples include warfarin and levothyroxine. These drugs require much tighter bioequivalence standards than standard medications.
How can I check if my drug is therapeutically equivalent?
The most reliable way is to consult the FDA's Orange Book or ask your pharmacist if the generic substitution is an "AB-rated" therapeutically equivalent product. Your pharmacist has access to these databases and can confirm the rating for you.
